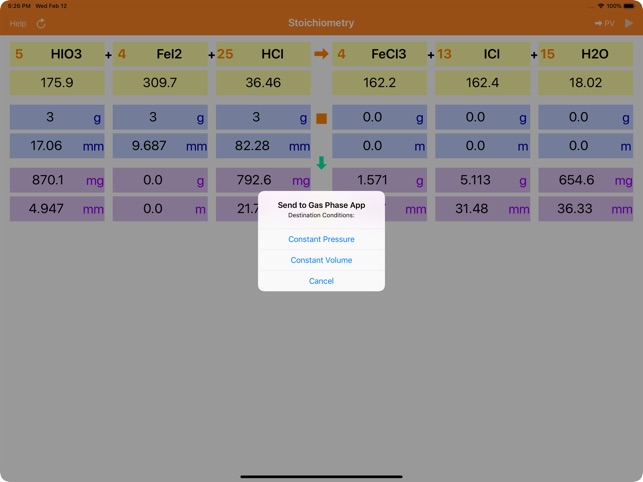
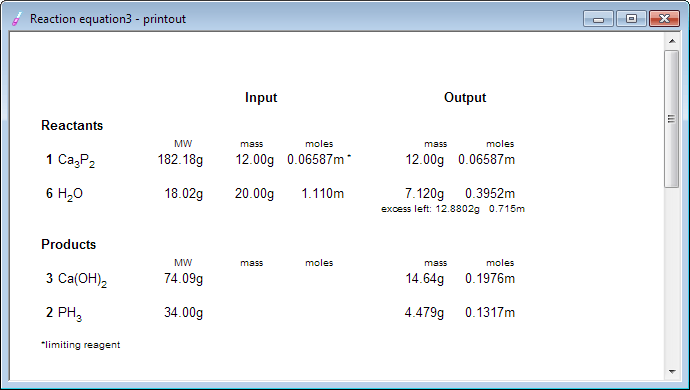
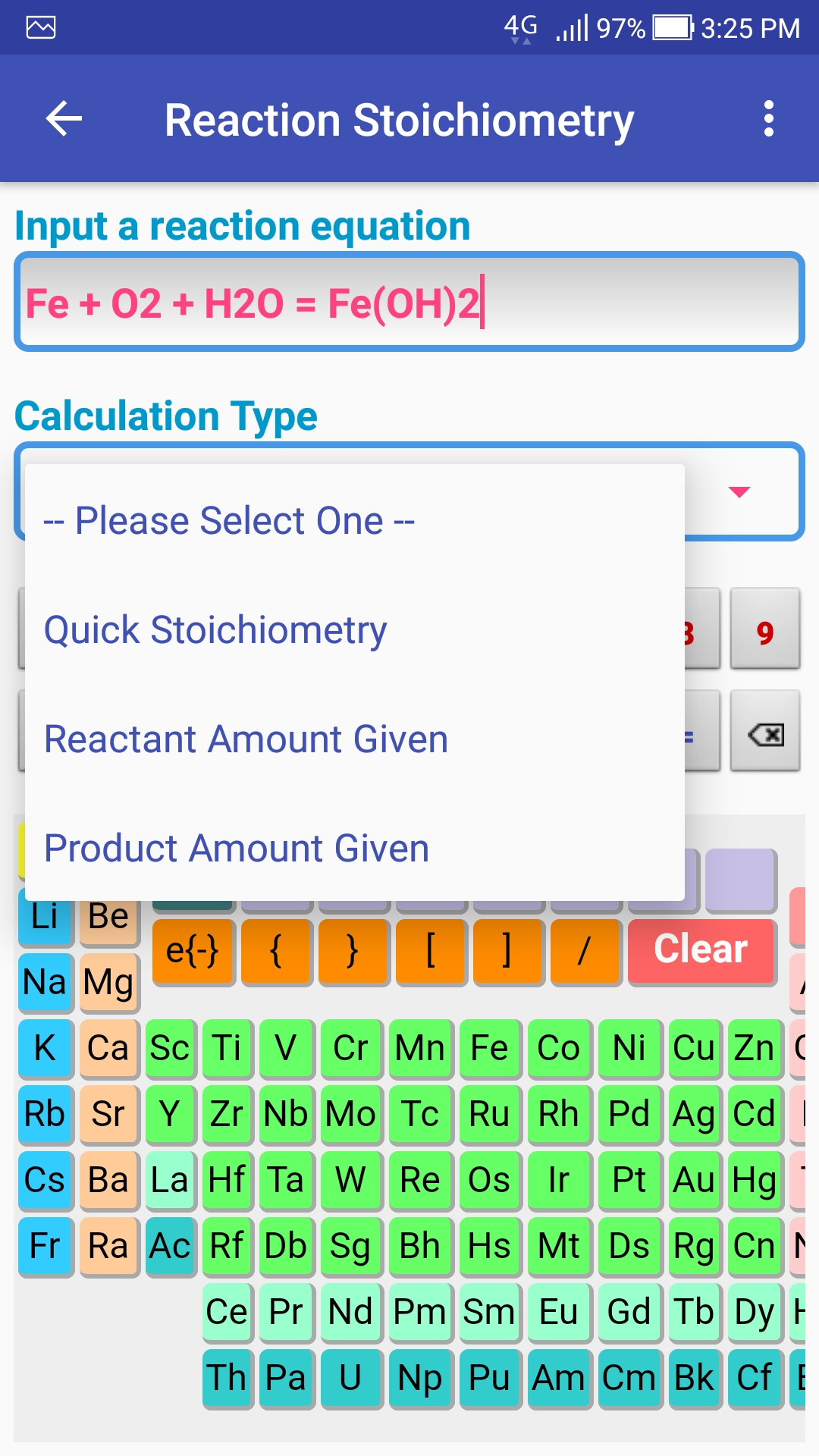
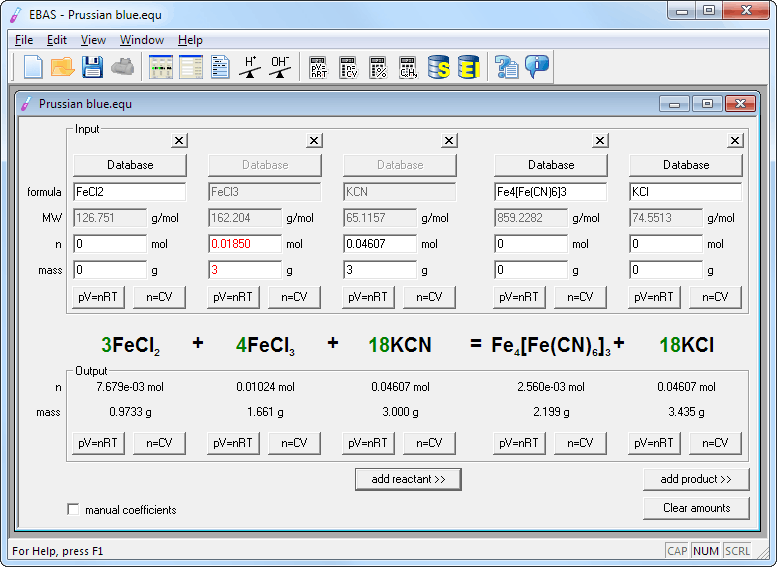
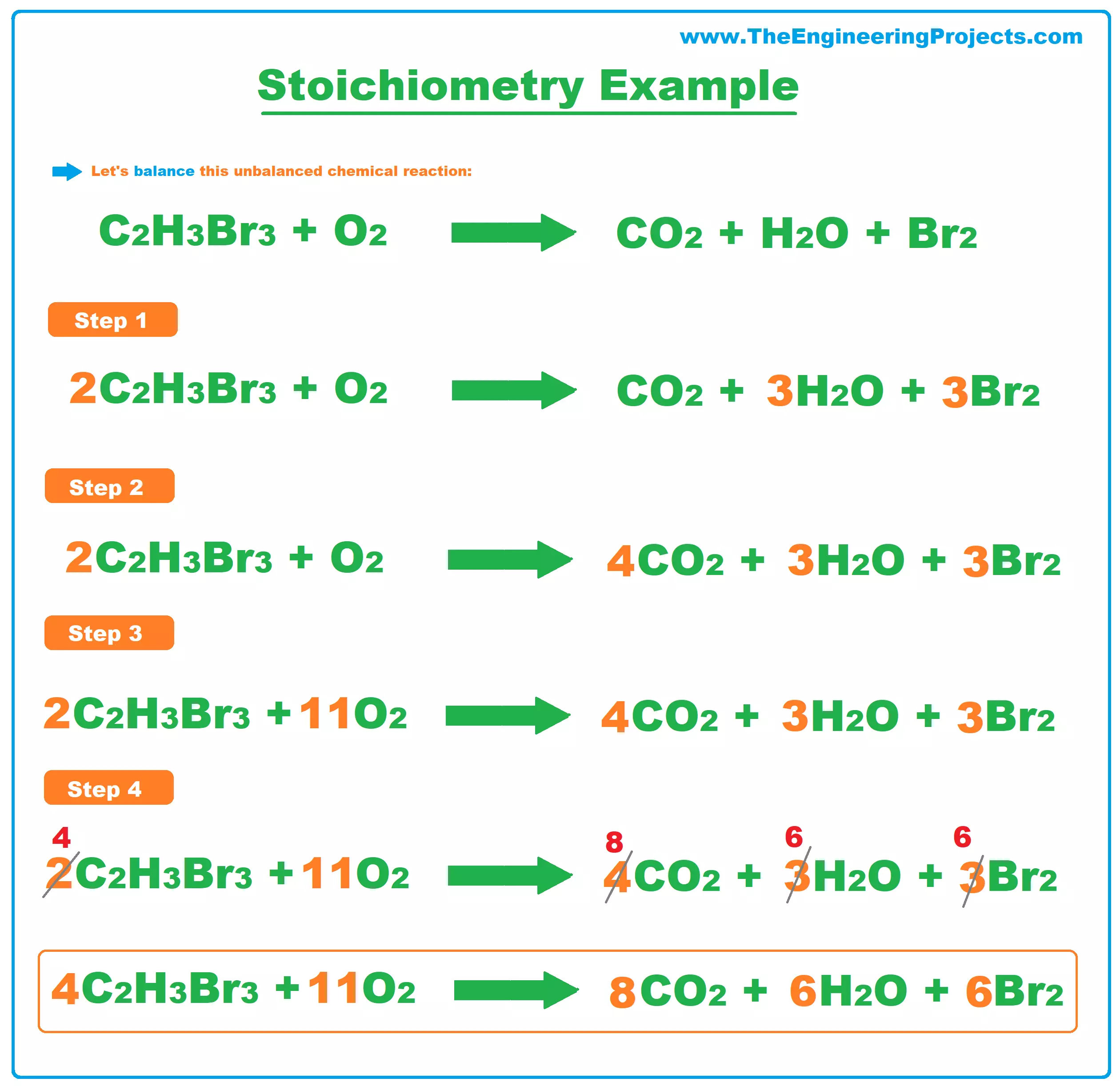
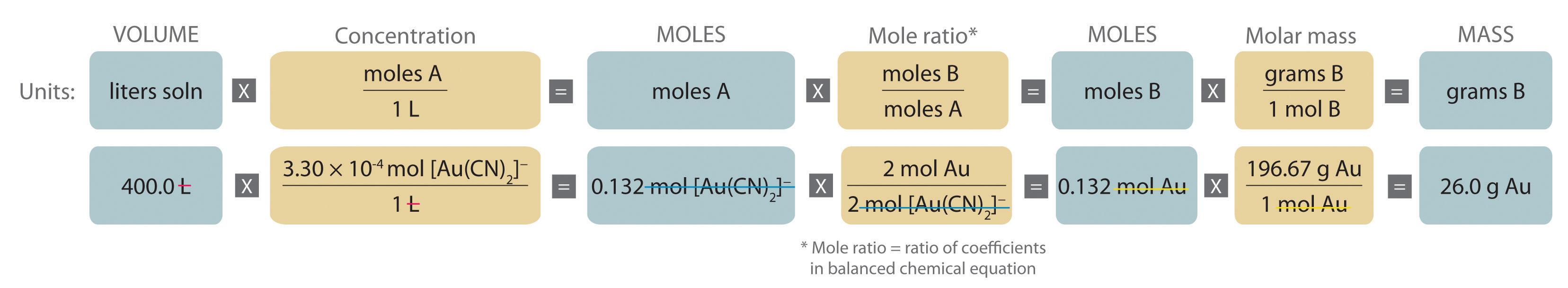
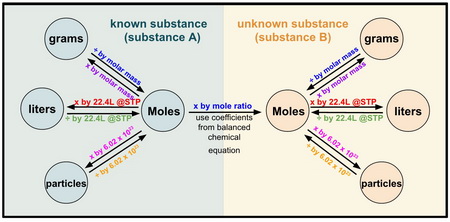
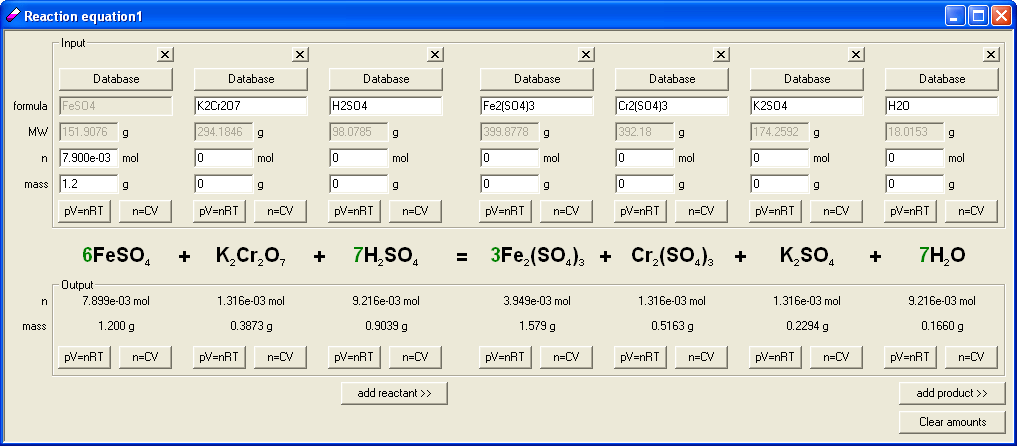
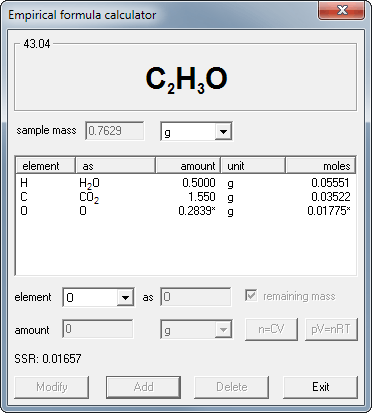
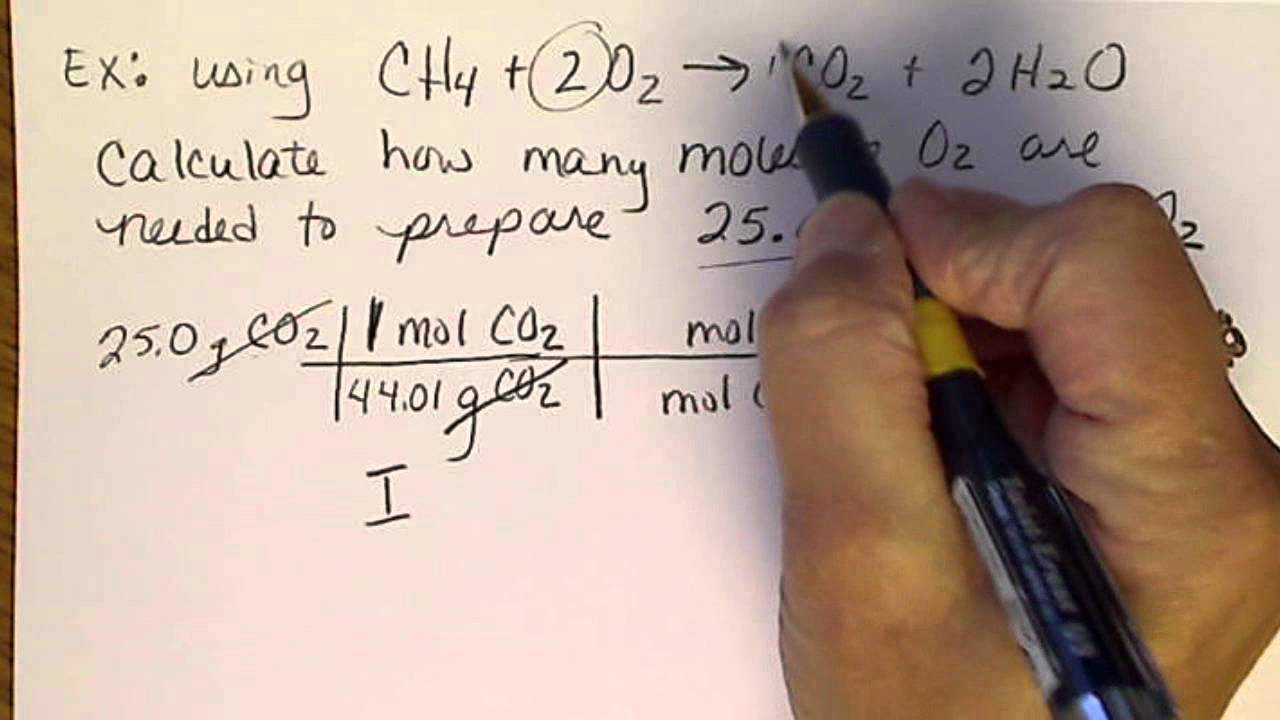EBAS stoichiometry calculator in action | Another chemicalforums question: A 25.0 mL sample of sulfuric acid is completely neutralized by adding 32.8 mL of 0.116 mol/L ammonia solution. Ammonium... | By ChembuddyFacebook

Solution Stoichiometry tutorial: How to use Molarity + problems explained | Crash Chemistry Academy - YouTube
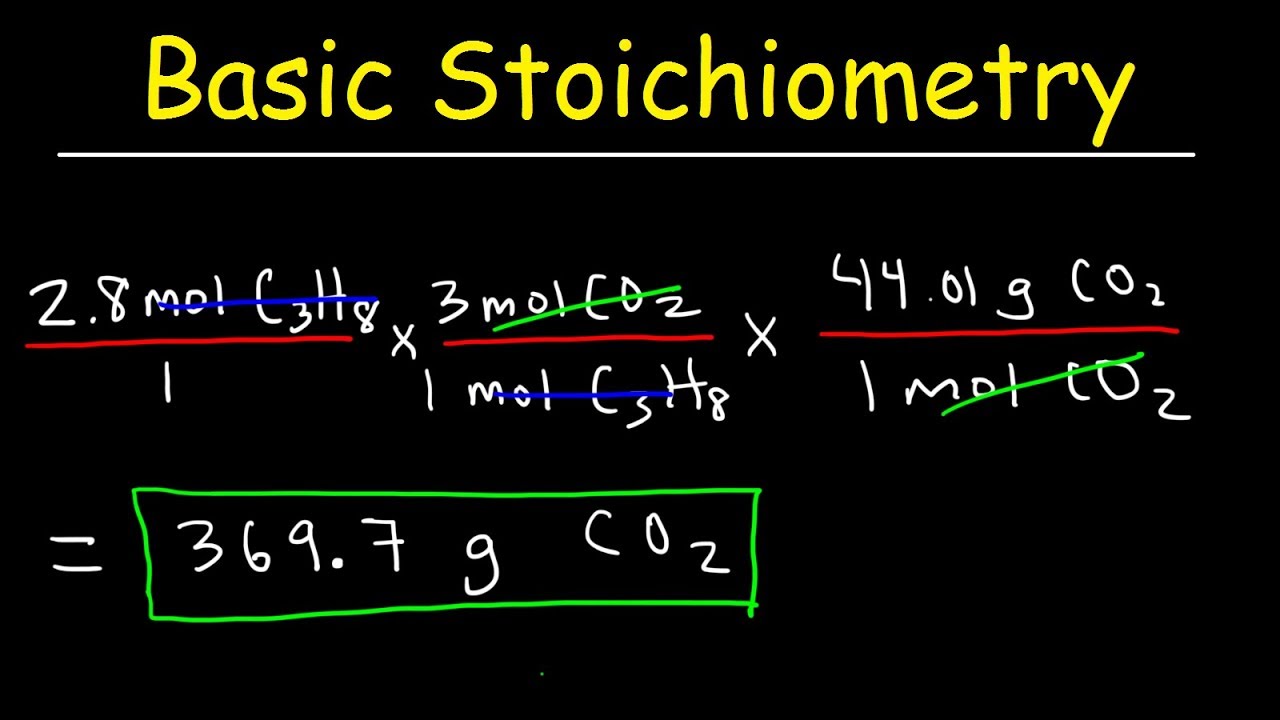
Stoichiometry Basic Introduction, Mole to Mole, Grams to Grams, Mole Ratio Practice Problems - YouTube
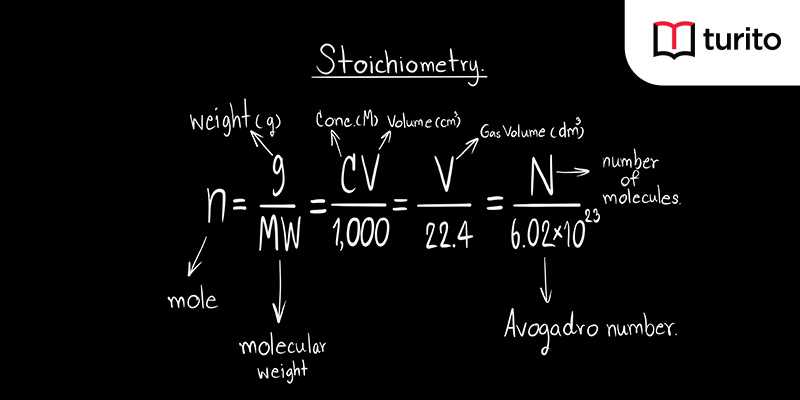
Stoichiometry Tutorial: Step by Step Video + review problems explained | Crash Chemistry Academy - YouTube