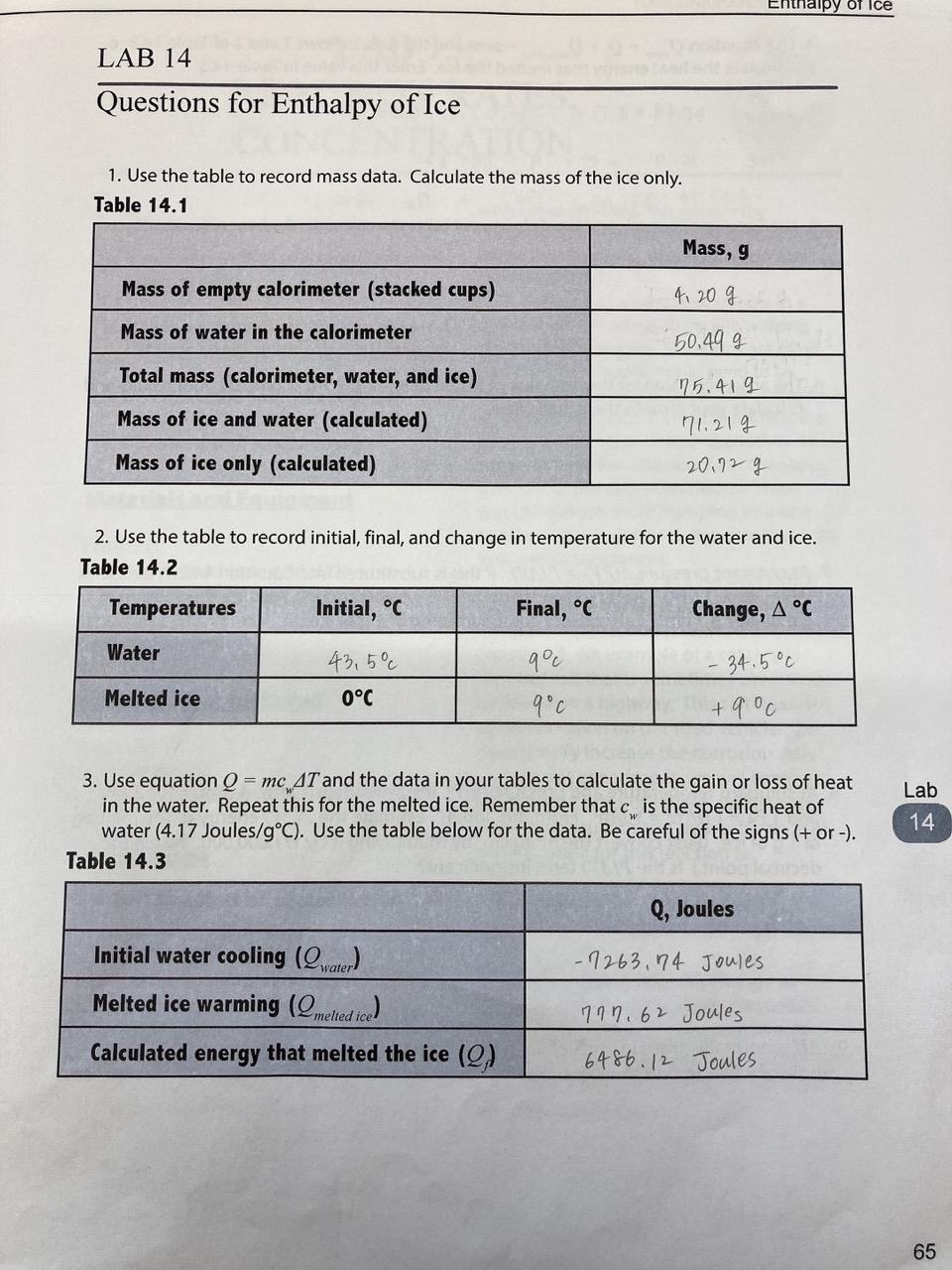
How to calculate the mass of ice used in a change of state experiment when I had 50g of water, an 80J calorimeter, an initial temperature of 46C and a final temperature

Specific Heat, J/g.°C 2.06 - ice 4.18 - water 2.03 - steam.Molar heat of fusion for water, kJ/mol - 6.02 - Home Work Help - Learn CBSE Forum

Question Video: Finding the Specific Latent Heat of fusion of Water given Its Mass, the Rate of Energy Transfer, and the Time Taken to Change State | Nagwa

Calculate the enthalpy change on freezing of `1 mol` of water at `10^(@)C` to ice at `-10^(@)C`. - YouTube
Enthalpy of Fusion and Vaporization of ice, water and vapour at room... | Download Scientific Diagram

SOLVED: Use standard enthalpies of formation to calculate the standard change in enthalpy for the melting of ice. (The H f for H2O(s) is -291.8 kJ>mol.) Use this value to calculate the
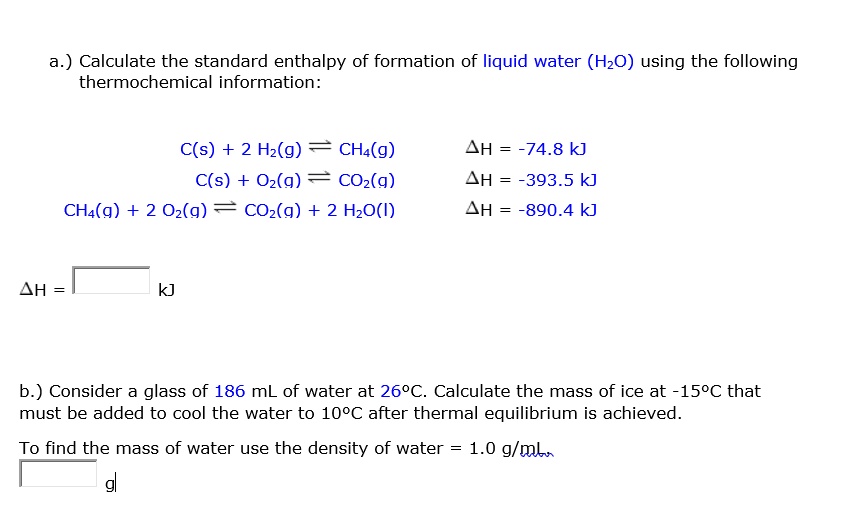
SOLVED: a.) Calculate the standard enthalpy of formation of liquid water (HzO) using the following thermochemical information: C(s) + 2 Hz(g) CHa(g) C(s) Oz(g) COz(g) CHa(g) + 2 Oz(g) COz(g) + 2


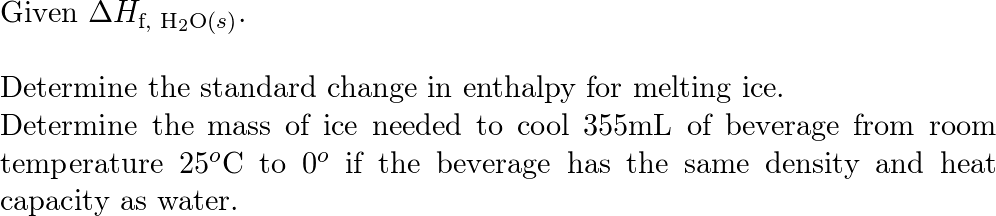

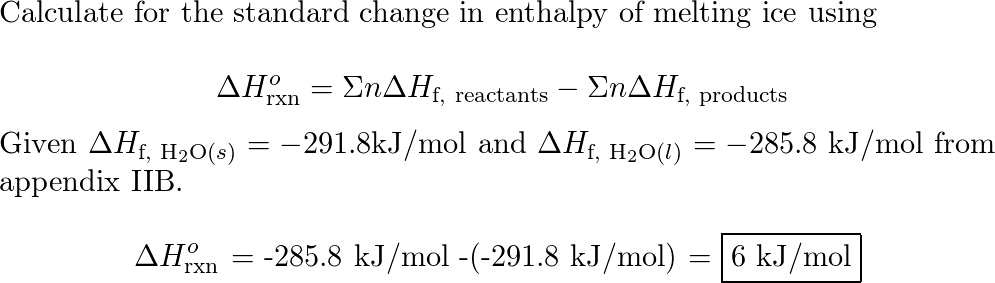






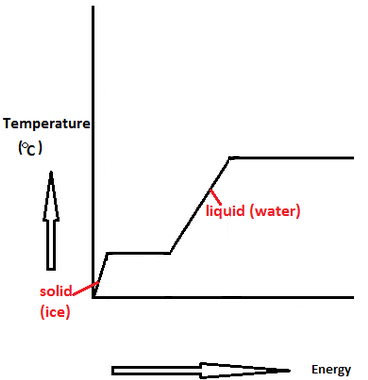
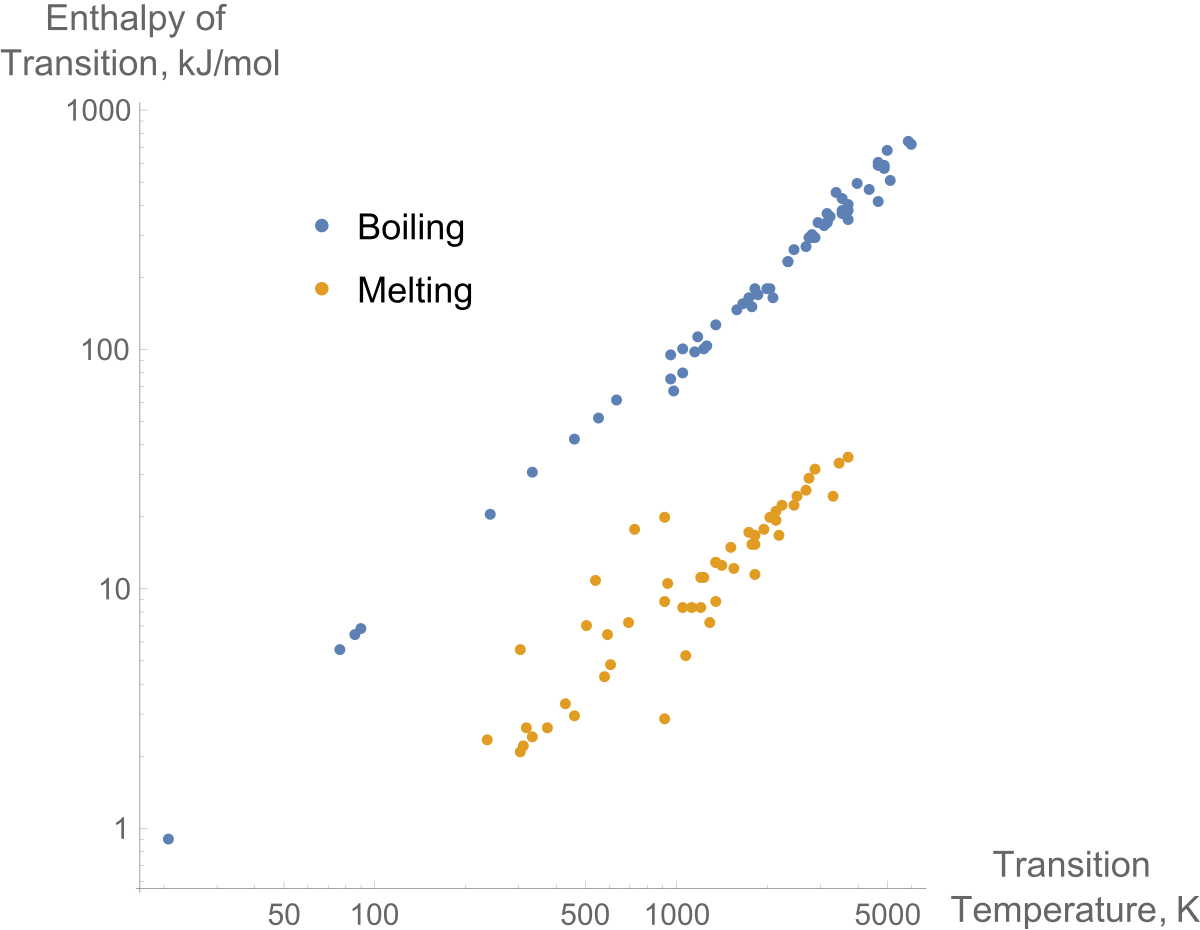

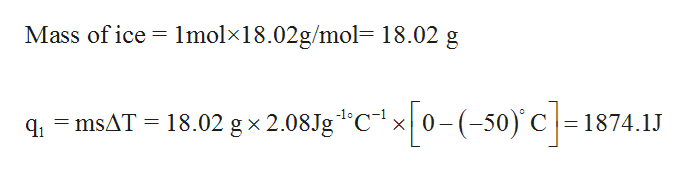
:max_bytes(150000):strip_icc()/GettyImages-1070123348-c9a136e91e7f4b6f9848581aa28b26d1.jpg)
