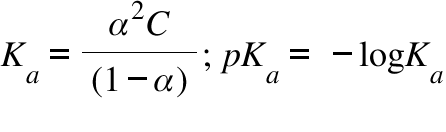Determination of the dissociation constant (K d ) of the full-length... | Download Scientific Diagram

A certain weak acid has dissociation constant of 1.0 × 10^-4 . The equilibrium constant for its reaction with a strong base is:

Dissociation Constants of Perchloric and Sulfuric Acids in Aqueous Solution | The Journal of Physical Chemistry B

equilibrium - How to calculate the dissociation constant of a weak acid from the titration with a strong base? - Chemistry Stack Exchange
The molar conductivity of 0.025 mol L-1 methanoic acid is 46.1 S cm2 mol-1 . Calculate its degree of dissociation and dissociation constant. - Sarthaks eConnect | Largest Online Education Community

Dissociation Constants of Perchloric and Sulfuric Acids in Aqueous Solution | The Journal of Physical Chemistry B

equilibrium - How to calculate the dissociation constant of a weak acid from the titration with a strong base? - Chemistry Stack Exchange